Optimization of Phenolic Compound Analysis in Marine Sediments through Improvement of USEPA Methods
Abstract
Phenolic compounds are generated from various industrial activities and natural processes, accumulating in marine sediments where they pose significant threats to ecosystems and human health. Despite their high toxicity and low environmental persistence, these compounds can accumulate at high concentrations over short periods, contributing to ongoing environmental contamination. This study improves and optimizes existing analytical methods, including USEPA 8270D, 3540C, and 3630C, for the quantitative analysis of phenolic compounds in marine sediments. To address limitations in the purification step of the analysis, experiments with various solvents were conducted. The 50% dichloromethane/ethyl acetate solvent exhibited the highest average recovery rate (85%) among the six mixed solvents tested, confirming its suitability as the optimal mixed solvent for purification in the simultaneous analysis of five phenolic compounds. In addition, quality assurance and quality control (QA/QC), including method detection limit (MDL), limit of quantification (LOQ), accuracy, and precision, were conducted to verify the feasibility of analyzing trace levels of phenolic compounds in sediments. The established analytical method was applied to coastal sediments from Busan and Ulsan in Korea, showing phenol as the major phenolic compounds. This method can be used to evaluate long-term accumulation trends of phenolic compounds in the marine environment and to establish effective pollution management strategies. The findings of this study provide reliable foundational data on phenolic compounds for monitoring marine pollution and establishing regulatory standards.
초록
페놀류는 다양한 산업 활동과 자연적 요인에 의해 생성되며, 해양환경 퇴적물에 축적되어 생태계와 인간 건강에 유해한 영향을 미친다. 이러한 화합물은 독성이 강하고 환경 중 잔류성이 낮음에도 불구하고, 단기간에 고농도로 축적될 수 있어 지속적인 환경 오염의 주요 원인이 된다. 본 연구는 해양환경 퇴적물 내 페놀류의 정량적 분석을 위해 기존 USEPA 8270D, 3540C, 3630C 분석법을 개선하고 최적화된 분석법을 제시한다. 분석의 정제 단계에서의 한계를 보완하기 위해 다양한 용매를 시험하였으며, 그 결과 여섯 가지 혼합용매 중 50% 디클로로메탄/에틸아세테이트가 평균 회수율 85%로 가장 우수하여 5종 페놀류 동시 분석의 정제 단계에 최적의 혼합 용매임을 확인하였다. 또한 방법검출한계(MDL), 정량한계(LOQ), 정확도, 정밀도를 포함한 QA/QC를 수행하여 퇴적물 중 미량 페놀류 분석의 타당성을 검증하였다. 확립된 분석법을 부산과 울산 연안 퇴적물에 적용한 결과, 페놀이 주요 페놀류로 확인되었다. 이 방법은 해양 환경에서의 페놀류 장기 축적 경향 평가와 효과적인 오염 관리 전략 수립에 활용될 수 있으며, 본 연구의 결과는 해양 오염 모니터링과 규제 기준 마련을 위한 신뢰성 높은 자료를 제공할 수 있게 되었다.
Keywords:
Phenolic compounds, Marine environment, Sediment, Gas chromatography/mass spectrometer, Analytical method키워드:
페놀류 화합물, 해양 환경, 퇴적물, 가스크로마토그래피/질량분석기, 분석법1. Introduction
Phenol is a chemical compound with a hydroxy group (-OH) attached to a benzene ring. When various substituents, such as nitro groups, methyl groups, and chloro groups, are added to phenol, they form an extensive range of derivatives, collectively known as phenolic compounds, numbering in the hundreds of thousands (Michalowicz and Duda [2007]; Lee et al. [1990]). In 2018, approximately 250 tons of phenolic compounds were released into the atmosphere and waterways in South Korea, with steadily increasing volumes released each year (National Institute of Environmental Research [2021]). Intensive industrial activities from petrochemical, plastic, pulp, and paper industries have released large amount of phenolic compounds into coastal areas (Dachs and Mejanelle [2010]). Once in the marine environment, phenolic compounds can increase the biological oxygen demand (BOD) of water under specific conditions, potentially exhibiting toxic effects on aquatic organisms depending on their concentration and exposure condition. The hydrophilic and biodegradable characteristics of phenolic compounds limit their bioaccumulation potential However, previous studies reported concerns for sensitive species, such as Euglena agilis and Scenedesmus quadricauda to indirect influence via food chains (Kottuparambil et al. [2014]; Wolff et al. [2015]; Davi and Gnudi [1999]; Tišler and Zagorc-Končan [1995]). Moreover, phenolic compounds exhibit adverse health effects on humans, such as carcinogenicity and disturbance of thyroid function, depending on their substituents (Chen et al. [2017]; Garabedian et al. [1999]; Honda and Kannan [2018]; Hsu et al. [2019]; Igbinosa et al. [2013]; Peng et al. [2013]; Szafran [2023]; Uberoi et al. [1997]).
Phenolic compounds have been detected in aquatic environments across multiple countries, including Spain, Australia, and Germany (Faludi et al. [2015]; El-Naggar et al. [2022]; Jauregui and Galceran [1997]; Jennings et al. [1996]; Piperidou et al. [1996]; Bolz et al. [2001]). Over time, these compounds can disperse and accumulate in sediments, potentially disrupting the functional integrity of aquatic ecosystems (Zaghloul et al. [2024]). While phenols exhibit short half-lives in water (9 days) and sediment (23 days), chemical spills or wastewater discharges may result in exceptionally elevated concentrations in sedimentary environments. The continuous input of phenolic compounds results in prolonged exposure and can have significant impacts on coastal ecosystems (Lee and Choi [2022]). For instance, various phenolic compounds, such as 2-nitrophenol (2-NP), 2,4-dichlorophenol (2,4-DCP), 2,4-dimethylphenol (2,4-DMP) and 2,4,6-trichlorophenol (2,4,6-TCP) have reported in sediments through case studies from Germany, Sweden, China, and Egypt (Reuneke et al. [2006]; Khairy [2013]; Wang et al. [2020]).
The U.S. Environmental Protection Agency (USEPA) has designated 11 phenolic compounds, including phenol itself, 2-chlorophenol, and 2-nitrophenol, as priority substances due to their high toxicity (USEPA [2014]). Similarly, the European Union classifies these compounds as substances of very high concern (SVHC) under the Registration, Evaluation, Authorization, and Restriction of Chemicals (REACH) regulation, while Japan manages them as priority assessment chemicals (PAC) under its Chemical Substance Control Law. In South Korea, phenol and pentachlorophenol are regulated as specific water pollutants under the Enforcement Decree of the Framework Act on Environmental Policy. Furthermore, several domestic studies have reported the detection of other phenolic compounds in freshwater, suggesting potential contamination of diverse phenolic compounds in Korean coastal sediments (Ko et al. [2007]; Park et al. [2023]). However, no analytical standards for phenolic compounds in sediments have been established in Korea, highlighting a regulatory gap compared to other countries.
Therefore, this study aims to develop an optimized analytical method for accurately quantifying phenolic compounds in marine sediments. Based on the USEPA methods 8270D, 3540C, and 3630C, the proposed method incorporates extensive literature review and experimental refinement to ensure its suitability for local conditions. To validate the method, QA/QC protocols were applied to ensure its reliability and reproducibility. Subsequently, the method was tested on sediment samples from Busan and Ulsan regions known for high pollution levels to assess its applicability in practical field conditions. The findings of this research provide critical insights into phenol contamination in marine sediments and offer a robust analytical tool for environmental monitoring and management.
2. Materials and Methods
2.1 Reagents and Target Compounds
In this study, five phenolic compounds were selected as target analytes, namely phenol (Phe), 2-nitrophenol, 2,4-dichlorophenol, 2,4-dimethylphenol, and 2,4,6-trichlorophenol, based on the priority list established by the United States Environmental Protection Agency (USEPA). A standard solution containing 2-chlorophenol-d4 and 2,4-dimethylphenol-d3 was used as the surrogate standard (SSTD), and 1,2-dimethyl-3-nitrobenzene was used as the gas chromatography/internal standard (GC/IS). All standard reagents were purchased from AccuStandard, diluted with acetone, and used for analysis. Solvents such as dichloromethane, ethyl acetate, and acetone, which were used in the extraction and purification processes, were of analytical grade or higher and were purchased from Honeywell.
2.2 Analytical Procedures
For the analysis of phenolic compounds in sediments, the extraction and purification processes were conducted following USEPA Methods 8270D, 3540C, and 3630C, with minor modifications as described in Section 2.2.2. Phenol, 2-nitrophenol, 2,4-dimethylphenol, 2,4-dichlorophenol, and 2,4,6-trichlorophenol were selected as target compounds based on their reported occurrence in previous studies of aquatic environments and their analytical feasibility using gas chromatography/mass spectrometry (GC/MS) (Ko et al. [2007]; Park et al. [2023]). To establish the analytical method, 20 g (wet weight) of sediment collected from Bijin Island, regarded as a non-contaminated area, was placed into a mortar and pestle with an appropriate amount of anhydrous sodium sulfate and homogenized until completely dried. The homogenized sample was placed in a thimble and subjected to Soxhlet extraction for 16 hours using a 50% acetone (ACT)/dichloromethane (DCM) mixed solvent. The extract was concentrated to 5 mL using a rotary evaporator. Subsequently, a purification process was performed using a column packed with 20 g of silicagel and 10 g of anhydrous sodium sulfate, with 15% toluene/hexane as the solvent. The purified solvent was further concentrated using a rotary evaporator and then reduced to 1 mL with high-purity nitrogen gas. Finally, instrumental analysis was performed using gas chromatography-mass spectrometry (GC/MS).
This study valuated the recovery rates of standard-spiked sediment samples extracted with six solvents, comprising both single and mixed types (40% toluene/hexane, 15% 2-propanol/toluene, methanol (MeOH), dichloromethane (DCM), hexane (HEX), and 50% ethyl acetate (ETAC)/dichloromethane (DCM)). These solvents included those used in the purification process of the USEPA method 3630C, applied to freshwater sediments, and those identified through a literature review as suitable for the analysis of phenolic compounds. The analysis was conducted using the same purification method with a total of six solvents. Through this process, the optimal purification solvent for the analysis of phenolic compounds in sediments was identified.
2.3 Instrumental Conditions
The equipment used for the analysis was a GC-MS (Agilent 7890B gas chromatography/5977B mass spectrometry), and the instrumental analysis conditions are shown in Table 1. The column used was a DB-5MS (30 m ×0.25 mm ×0.25 μm film thickness), and high-purity helium (99.999%) was used as the inert carrier gas. The carrier gas flow rate was set to 1.5 mL/min. The GC conditions were set with an injection port temperature of 275°C, and the sample was introduced in splitless injection mode. The oven temperature was programmed to start at 60°C, then increase to 300°C at a rate of 8°C/min, with a hold time of 10 minutes. The mass spectrometer (MS) conditions were set with a transfer line temperature of 280°C, and the electron impact (EI) ionization mode was used. The ion source temperature was set to 230°C. Selective ion monitoring (SIM) was employed as the detection method, and the solvent delay time was set to 5 minutes to prevent damage to the instrument. The retention times (RT) and quantification/qualification ions of the target compounds are presented in Table 2. Quantification was performed using GC/MS, and the concentrations were corrected based on surrogate recovery.
2.4 Quality Assurance and Quality Control (QA/QC)
In this study, the method detection limits (MDLs), limits of quantitation (LOQs), accuracy, and precision were calculated to ensure the reliability and accuracy of the analysis results. For all target compounds, the limits of quantifications (LOQs) were determined using solvent spiked at 1 ng/mL. The method detection limits (MDLs) were obtained using spiked sediment samples at 1 ng/g ww, whereas the accuracy and precision were evaluated using sediment samples spiked at 500 ng/g ww. Seven replicate analyses were performed using phenol standard solutions at the lowest concentration level. The MDL was calculated by multiplying the standard deviation (σ) of the replicate measurements by the t-value of 3.14, corresponding to the 99% confidence level. The LOQ was determined by multiplying the standard deviation by a factor of 10, representing the threshold for reliable quantification. Accuracy was calculated by dividing the measured concentration by the actual concentration of the standard material and expressing it as a percentage. Precision was calculated by dividing the standard deviation of the analysis results for the 7 replicate samples by the average value and expressing it as a percentage.
2.5 Field Application
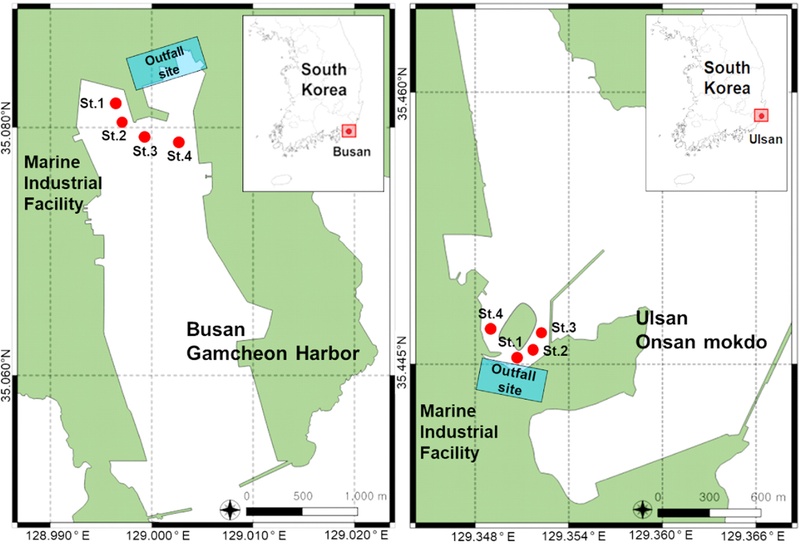
To validate the optimized method, sediment samples were collected from four sites near marine industrial facilities in Busan (St. 1 ~ 4) and Ulsan (St. 1 ~ 4) (Fig. 1). To prevent phenol contamination during sediment sample collection, aluminum containers were used, which were cleaned with dichloromethane and completely dried before sampling. The collected samples were immediately transferred to aluminum containers, double-bagged in zipper bags, and stored. Furthermore, to minimize photochemical degradation of phenolic compounds, the samples were stored in a dark place (Chowdhury et al.[2017]) and frozen at -20°C until analysis. Before analysis, the samples were thawed at room temperature, homogenized, and then used. Each field sample was analyzed in duplicate. To evaluate potential background contamination during the experimental procedures, two types of blank samples were prepared. The first was composed of uncontaminated marine sediment, and the second consisted of anhydrous sodium sulfate, selected for its similar physical characteristics to sediment. Both blanks were subjected to the same procedures as the field samples, including sampling, storage, pretreatment, and instrumental analysis. These blanks were used to assess potential contamination introduced during the entire experimental process.
3. Results and Discussion
3.1 Problems with Existing Analytical Methods
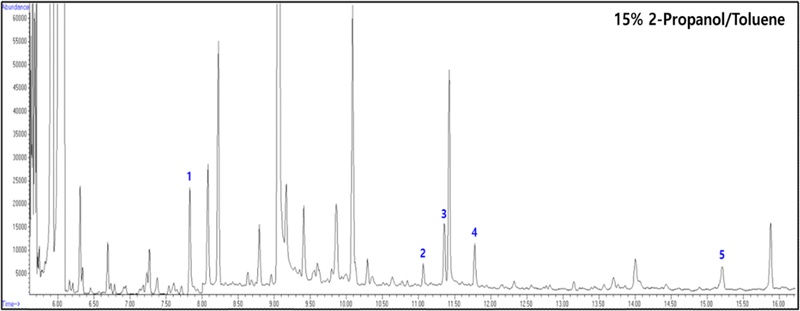
Based on USEPA Methods 8270D, 3530C, and 3630C, sediment samples in previous studies were extracted using Soxhlet extraction and subsequently purified using 15% 2-propanol in toluene as the final cleanup solvent. GC/MS analysis revealed several issues with the toluene solvent during both the concentration process and chromatogram analysis. During the rotary evaporation concentration process, the high boiling point of toluene made it difficult to set appropriate concentration conditions. In the nitrogen concentration process, it took more than three times as long compared to other organic solvents such as dichloromethane or hexane. In the chromatogram, numerous impurity peaks unrelated to the target analytes were observed, which complicated data interpretation (Fig. 2). These issues clearly highlight the problems with the existing purification process and emphasize the need for improvement.
3.2 Establishment of the Optimal Purification Solvent
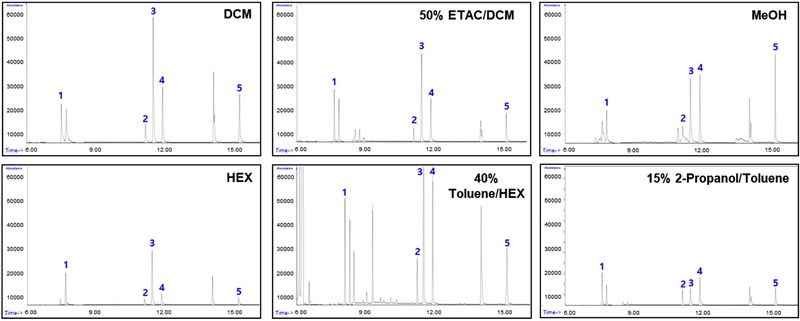
To improve the purification process, the recovery rates and chromatograms of five solvents—DCM, MeOH, HEX, 40% toluene/HEX and 50% ETAC/DCM —were further evaluated, in addition to 15% 2-propanol/toluene (Table 3, Fig. 3). The 50% ETAC/DCM showed the highest average recovery rate of over 85%, with almost no impurity peaks observed in the chromatogram. MeOH showed high recovery rates for Phe and 2,4-DMP, but lower rates for 2,4-DCP and 2,4,6-TCP. Among the various solvents tested, the methanol experiments yielded recoveries that often far exceeded 100%. These results may be due to the high polarity of methanol, which facilitates the co-extraction and elution of various polar background constituents in the sediment. This overlap with the target analyte peaks can consequently lead to an overestimation of the instrumental response. HEX showed good recovery for the surrogate standard but had low recovery rates for all target compounds, with the lowest recovery observed for 2,4,6-TCP. The 40% toluene/hexane solvent showed good recovery for specific compounds, but the recovery of the surrogate standard was relatively low, and multiple impurity peaks were observed in the chromatogram. The 15% 2-propanol/toluene solvent exhibited excellent recovery for certain compounds, but overall analytical efficiency was lower compared to 50% ETAC/DCM. Ultimately, 50% ETAC/DCM was selected as the purification solvent to maximize experimental efficiency.
3.3 Quality assurance and quality control
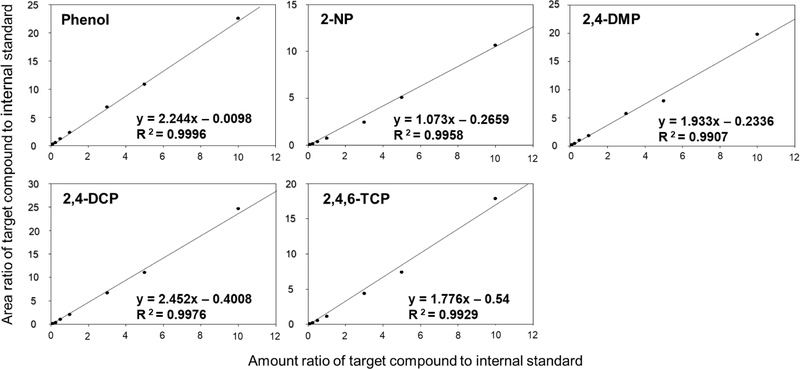
In this study, quality assurance and quality control procedures were implemented to evaluate the reliability and accuracy of the analysis for the following five phenolic compounds, confirming their applicability. Standard solutions were prepared within a concentration range of 10–1000 μg/L and analyzed using GC/MS to create calibration curves. The determination coefficients (R2) for all compounds were confirmed to be above 0.99 (Fig. 4).
In this study, the detection limits, quantification limits, accuracy, and precision were calculated to evaluate the reliability and accuracy of the analysis (Table 4). The method detection limits ranged from 0.005 to 0.028 ng/g for all compounds, confirming that even trace amounts of phenolic compounds can be detected. The limits of quantification ranged from 0.017 to 0.090 ng/g, demonstrating that quantification is possible even in actual environmental samples. In addition, solvent and method blanks were analyzed with each extraction batch, and all target analytes were below the detection limit. The accuracy ranged from 95% to 135% for all compounds, meeting the criteria of 65% to 135% set by USEPA method 8040. The precision values ranged from 3.1% to 5.6%, falling within the recommended value of 30% by EPA method 8040. These results confirm that the analytical method established in this study is reliable and reproducible for the analysis of phenolic compounds in sediments.
3.4 Field Application
In the Gamcheon Harbor, the concentration of Phe ranged from 28.2 ng/g to 47 ng/g, while in the Onsan Bay, it ranged from 105.7 ng/g to 825.9 ng/g (Table 5). These values are similar to the Phe concentrations (30–330 ng/g) reported in freshwater sediments in other countries (Abdallah and Alprol [2024]; Kim et al.[2023]; Zhou et al.[2017]). In both Gamcheon Port in Busan and Onsan Bay in Ulsan, Phe accounted for more than 95% of the phenolic compounds at all sampling points, which is consistent with previous studies where Phe comprised over 90% of phenolic compounds in most sediments (Kim et al. [2023]). In the Onsan Bay near marine industrial facilities, the highest concentration of Phe was found at 825.9 ng/g dw, indicating that the discharge point near St.1 is likely the source of the contamination. Overall, higher concentrations were observed near the Ulsan marine industrial area compared to the Busan area. Compared to Gamcheon Bay, Onsan Bay has larger scale industrial facilities. Furthermore, the sediments in Onsan Bay are finer-grained and contain higher levels of organic carbon than those in Gamcheon Bay (Back et al. [2016]). Other phenolic compounds were mostly undetectable, with only trace amounts detected in some areas.
4. Conclusion
In this study, an optimized analytical method was developed by improving the existing USEPA method 3630C for the analysis of phenolic compounds in marine sediments. Testing of various purification solvents revealed that the 50% ETAC/DCM achieved the highest recovery rates and stability, making it the optimal purification solvent. QA/QC results demonstrated high accuracy and precision, confirming the reliability of the method. Analysis of sediment samples from the coasts of Busan and Ulsan identified Phe as the predominant compound, with particularly high concentrations detected in the Ulsan region. The analytical method developed in this study provides sufficient sensitivity to measure phenolic compounds at levels present in Korean coastal sediments, thereby enabling the monitoring of their long-term behavior and contamination trends influenced by industrial activities.
Acknowledgments
This research was supported by Korea Institute of Marine Science & Technology Promotion (KIMST) funded by the Ministry of Oceans and Fisheries, Korea (Development of technology for impact assessment and management of HNS discharged from marine industrial facilities [RS-2021-KS211535], Development of risk managing technology tackling ocean and fisheries crisis around Korean Peninsula by Kuroshio Current [RS-2023-00256330]) and Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education [RS-2024-00413748].
References
-
Abdallah, M.A.M. and Alprol, A.E., 2024, Using phenolic compounds from the Mediterranean Sea west of Egypt to categorize the sediment quality, Discov. Mater., 4, 1-10.
[https://doi.org/10.1007/s43939-024-00158-7]

- Back, H., Kim, O., 2016, Development of ecosystem health index for the Southern Sea Special Management Area (Phase II, 3rd year), Project report submitted to the Korea Institute of Ocean Science and Technology (KIOST), Busan, Korea.
-
Bolz, U., Hagenmaier, H. and Korner, W., 2001, Phenolic xenoestrogens in surface water, sediments, and sewage sludge from Baden-Württemberg, south-west Germany, Environ. Pollut., 115, 291-301.
[https://doi.org/10.1016/S0269-7491(01)00100-2]

-
Bugg, T.D., Ahmad, M., Hardiman, E.M. and Rahmanpour, R., 2011, Pathways for degradation of lignin in bacteria and fungi, Nat. Prod. Rep., 28, 1883-1896.
[https://doi.org/10.1039/C1NP00042J]

-
Chen, R., Yuan, L., Zha, J. and Wang, Z., 2017, Developmental toxicity and thyroid hormone-disrupting effects of 2,4-dichloro-6-nitrophenol in Chinese rare minnow (Gobiocypris rarus), Aquat. Toxicol., 185, 40-47.
[https://doi.org/10.1016/j.aquatox.2017.02.005]

-
[6] Chowdhury, P., Nag, S. and Ray, A.K., 2017, Degradation of phenolic compounds through UV and visible-light-driven photocatalysis: technical and economic aspects, in: Soto-Hernandez, M., Palma-Tenango, M. and Garcia-Mateos, R. (Eds.), Phenolic Compounds - Natural Sources, Importance and Applications, InTech, Rijeka, 395–417.
[https://doi.org/10.5772/66134]

-
Dachs, J. and Mejanelle, L., 2010, Organic pollutants in coastal waters, sediments, and biota: a relevant driver for ecosystems during the anthropocene, Estuar. Coasts, 33, 1-14.
[https://doi.org/10.1007/s12237-009-9255-8]

-
Davi, M.L. and Gnudi, F., 1999, Phenolic compounds in surface water, Water Res., 33, 3213-3219.
[https://doi.org/10.1016/S0043-1354(99)00027-5]

-
El-Naggar, N.A., Moawad, M.N. and Ahmed, E.F., 2022, Toxic phenolic compounds in the Egyptian coastal waters of Alexandria: spatial distribution, source identification, and ecological risk assessment, Water Sci., 36, 32-40.
[https://doi.org/10.1080/23570008.2022.2031724]

-
Faludi, T., Balogh, C., Serfőző, Z. and Molnár-Perl, I., 2015, Analysis of phenolic compounds in the dissolved and suspended phases of Lake Balaton water by gas chromatography-tandem mass spectrometry, Environ. Sci. Pollut. Res., 22, 11966-11974.
[https://doi.org/10.1007/s11356-015-4734-x]

-
Garabedian, M.J., Hoppin, J.A., Tolbert, P.E., Herrick, R.F. and Brann, E.A., 1999, Occupational chlorophenol exposure and non-Hodgkin's lymphoma, J. Occup. Environ. Med., 41, 267-272.
[https://doi.org/10.1097/00043764-199904000-00008]

-
Honda, M. and Kannan, K., 2018, Biomonitoring of chlorophenols in human urine from several Asian countries, Greece and the United States, Environ. Pollut., 232, 487-493.
[https://doi.org/10.1016/j.envpol.2017.09.073]

-
Igbinosa, E.O., Odjadjare, E.E., Chigor, V.N., Igbinosa, I.H., Emoghene, A.O., Ekhaise, F.O., Nicholas, O.I. and Idemudia, O.G., 2013, Toxicological profile of chlorophenols and their derivatives in the environment: the public health perspective, Sci. World J., 2013, Article ID 460215.
[https://doi.org/10.1155/2013/460215]

-
Jauregui, O. and Galceran, M.T., 1997, Determination of phenols in water by on-line solid-phase disk extraction and liquid chromatography with electrochemical detection, Anal. Chim. Acta, 340, 191-199.
[https://doi.org/10.1016/S0003-2670(96)00504-1]

- Jeon, S.S., 2021, Monitoring of phenolic compound and cyanide in tree residues and soils as affected by forest fires, M.S. thesis, Dept. Food Environ. Converg., Kangwon Natl. Univ., Chuncheon, Korea.
-
Jennings, J.G., De Nys, R., Charlton, T.S., Duncan, M.W. and Steinberg, P.D., 1996, Phenolic compounds in the nearshore waters of Sydney, Australia, Mar. Freshw. Res., 47, 951-959.
[https://doi.org/10.1071/MF9960951]

-
Kim, D.H., Choi, S., Park, J., Kim, K. and Oh, J.E., 2023, Phenolic compounds in the freshwater environment in South Korea: occurrence and tissue-specific distribution, Sci. Total Environ., 905, 166914.
[https://doi.org/10.1016/j.scitotenv.2023.166914]

-
Khairy, M.A., 2013, Assessment of priority phenolic compounds in sediments from an extremely polluted coastal wetland (Lake Maryut, Egypt), Environ. Monit. Assess., 185, 441-455.
[https://doi.org/10.1007/s10661-012-2566-4]

-
Ko, E.J., Kim, K.W., Kang, S.Y., Kim, S.D., Bang, S.B., Hamm, S.Y. and Kim, D.W., 2007, Monitoring of environmental phenolic endocrine disrupting compounds in treatment effluents and river waters, Korea, Talanta, 73, 674-683.
[https://doi.org/10.1016/j.talanta.2007.04.033]

-
Kottuparambil, S., Kim, Y.J., Choi, H., Kim, M.S., Park, A., Park, J. and Han, T., 2014, A rapid phenol toxicity test based on photosynthesis and movement of the freshwater flagellate, Euglena agilis Carter, Aquat. Toxicol., 155, 9-14.
[https://doi.org/10.1016/j.aquatox.2014.05.014]

-
Lee, A. and Choi, Y., 2022, Effect of the fate mechanisms of phenol on the remediation efficiency of in-situ capping applied to sediment contaminated by phenol chemical spills, J. Soil Groundw. Environ., 27, 60-70.
[https://doi.org/10.7857/JSGE.2022.27.1.060]

- Michalowicz, J. and Duda, W., 2007, Phenols - sources and toxicity, Pol. J. Environ. Stud., 16, 3.
- National Institute of Environmental Research (NIER), 2021, Results of Pollutant Release and Transfer Register (PRTR) in 2021, National Institute of Environmental Research, Incheon, Korea.
- Park, S., Choi, J., Lee, J. and Kim, H., 2023, Occurrence and risk assessment of phenol and substituted phenols in water and fish collected from the streams in eastern Gangwon State, Korea, Anal. Sci., 36, 224-235.
- Piperidou, C.I., Chaidou, C.I., Saraci, M., Stalikas, C. and Pilidis, G., 1996, Determination of phenolic compounds, PAHs and heavy metals in surface waters in Albania, Fresenius Environ. Bull., 15, 557-562.
-
Tišler, T. and Zagorc-Končan, J., 1995, Relative sensitivity of some selected aquatic organisms to phenol, Bull. Environ. Contam. Toxicol., 54, 717-723.
[https://doi.org/10.1007/BF00206104]

-
Wang, X., Chen, A., Chen, B. and Wang, L., 2020, Adsorption of phenol and bisphenol A on river sediments: effects of particle size, humic acid, pH and temperature, Ecotoxicol. Environ. Saf., 204, 111093.
[https://doi.org/10.1016/j.ecoenv.2020.111093]

-
Zaghloul, G.Y., Mohamedein, L.I., Kelany, M.S., El-Moselhy, K.M. and Ezz El-Din, H.M., 2024, Impact of total phenolic compounds on ecological and health risks of water and sediments from Timsah Lake, Suez Canal, Egypt, Environ. Sci. Pollut. Res., 31, 45667-45682.
[https://doi.org/10.1007/s11356-024-34047-6]